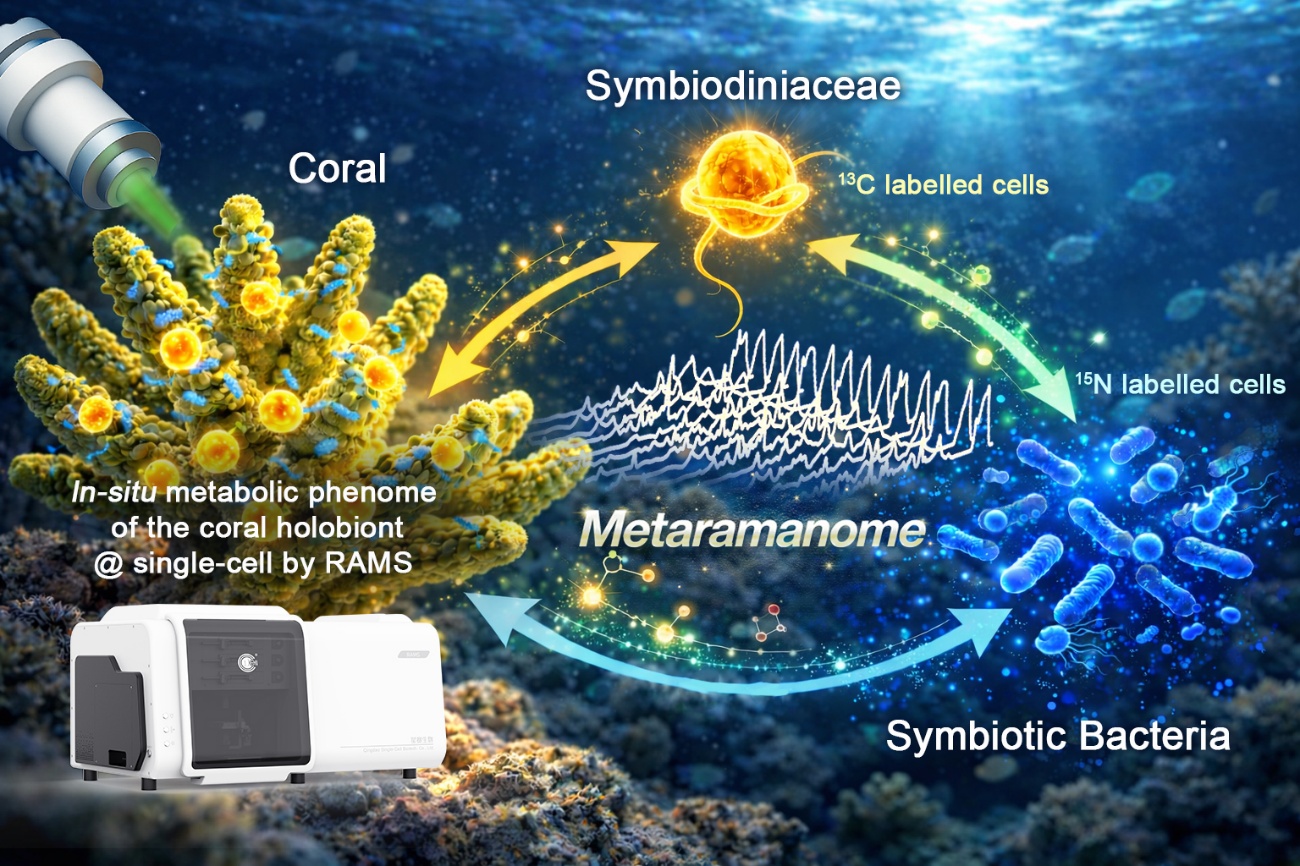
A recently completed study has successfully developed an innovative approach for in-situ detection of metabolites in single cells of corals. Supported by eCyte's RAMS (Raman-Activated Microfluidic Sorter) single-cell metabolic analysis platform, this research provides new insights into the transfer and recycling of carbon and nitrogen across the coral-Symbiodiniaceae-bacteria consortium, and has constructed the first single-cell, in-situ metabolic atlas of the coral holobiont.
Using the RAMS platform, the research team conducted metabolic phenotypic analysis of two Symbiodiniaceae strains (symbiotic clade C and non-symbiotic clade E) following long-term high-temperature acclimation, successfully generating thermotolerant strains. Through high-throughput single-cell metabolic fingerprinting enabled by RAMS, the researchers discovered pronounced metabolic reprogramming. Under heat stress, which typically depletes intracellular proteins, carbohydrates, and lipids, the thermotolerant strains exhibited enhanced protein- and lipid-associated signatures, with some cells accumulating starch, making them ideal for subsequent phenotypic tracking and targeted screening.
Guided by metagenomic screening and validated by in vitro assays, the team identified a symbiotic bacterium (Muricauda sp.) closely associated with both the coral host and Symbiodiniaceae. Using ¹³C and ¹⁵N substrate labeling in Symbiodiniaceae-bacteria co-cultures, and comparing single-cell Raman spectra before and after feeding via the RAMS platform, they quantified carbon-nitrogen exchange dynamics and uncovered partner-dependent transfer regimes. In the bacteria-clade E pairing, exchanged metabolites were dominated by amino acid-associated signatures. In contrast, exchange with clade C was more efficient, extending beyond amino acids and carbohydrates to include bidirectional transfer of nucleic acid-associated compounds. This finding suggests that exchange directionality and temporal ordering may be key determinants of symbiotic metabolic efficiency.
At the holobiont level, the RAMS platform successfully tracked metabolic remodeling during bleaching, revealing significant depletion of proteins, lipids, and carbohydrates in coral tissues as bleaching severity increased. The researchers then introduced ¹³C-labeled thermally acclimated Symbiodiniaceae together with ¹⁵N-labeled bacteria into bleached corals, using the RAMS platform to monitor metabolic phenotypic changes during recovery. Although there was little difference in short-term visible recovery among treatments, co-inoculation of clade C with bacteria significantly enhanced protein-, lipid-, and carbohydrate-associated signals in coral tissues, indicating partial mitigation of physiological impairment with stronger effects than either inoculum alone. Conversely, clade E inoculation resulted in carbohydrate signal accumulation. Microscopy and nucleic acid quantification supported the establishment of short-term symbiosis and altered host nutrient partitioning.
eCyte's RAMS single-cell metabolic analysis platform provided critical technological support for this study. Its capability for real-time monitoring of metabolic exchanges at single-cell resolution offers a novel research tool for understanding coral symbiotic mechanisms. The application of this platform further advances research exploration in microbial-host metabolic interactions and ecosystem conservation.






